AM for severe dry eye?
Despite different underlying pathogenic processes, inflammation is a common denominator in dry eye disease (DED), which in turn promotes further damage to the corneal epithelium and its underlying structures.
Several treatment modalities have been used to suppress inflammation, but results are variable and refractory in some cases. The use of amniotic membrane (AM) as another treatment modality in DED is, however, very exciting.
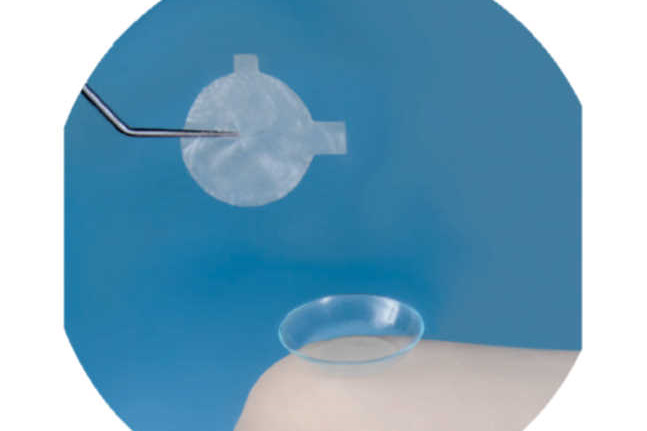
AM is part of the amnion and consists of a single avascular layer of human-processed epithelial cells, with a basement membrane. Its efficacy is attributed to its intrinsic growth factors which promote healing and its potent anti-inflammatory effect. Biotherapeutic AM can be either transplanted in theatre, with sutures or glue, or it can be used in office. It comes in two forms: cryopreserved amniotic membrane or, more recently, a sterile, dehydrated AM (Amnio-Tek) with a three-year, room temperature shelf life. Both forms can be used in office.
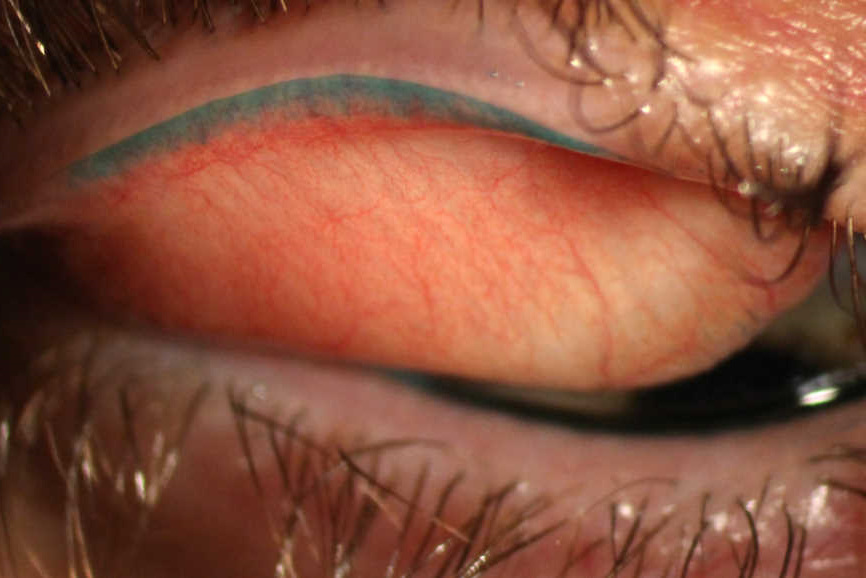
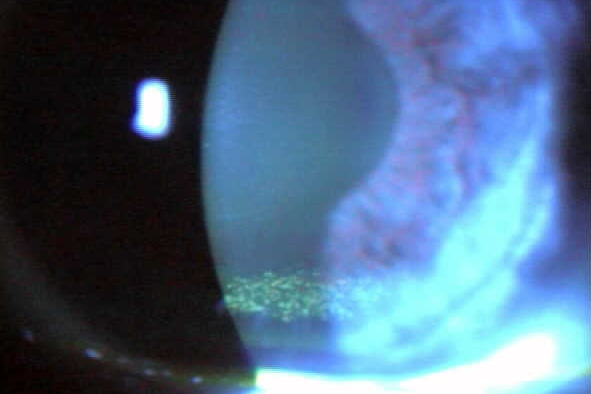
We have recent experience of applying AM (in its dehydrated form) in a severe dry eye case with persistent epithelial defect that failed to respond to other modalities. It adheres by capillary action and a bandage contact lens on top helps keep it in place and protect it.
The short-term outcome has been favourable, however, long-term follow up is required to assess the efficacy of AM in severe dry eye disease.
Dr Rasha Altaie is a consultant ophthalmologist with Counties Manukau DHB and Milford Eye, with specialist knowledge in cornea and anterior segment disease.