Getting to grips with RPD and AMD
Late last year a world-leading study was launched concentrating specifically on reticular pseudodrusen (RPD) and why they appear to be a high-risk phenotype for age-related macular degeneration (AMD) progression.
A team led by principal investigator Professor Robyn Guymer, deputy director and head of macular research at the Centre for Eye Research Australia (CERA), instigated a large multidiscipline effort to explore why some patients with the earliest stages of AMD have a higher risk of rapid progression to permanent vision loss.
With the extensive prevalence of AMD across the globe, Prof Guymer’s study involves multiple, national and international experts and analysis of tens of thousands of patient scans. There is no effective treatment for dry AMD or proven ways to slow progression from early to late AMD. There is also uncertainty around why some people with AMD are more at risk of losing their sight than others. Thus, the research aims to discover the predisposing factors for rapid progression to vision loss in the ‘high-risk’ AMD patient with RPD. The pathophysiology of AMD is complex and there is currently much to discover about the underlying cause of RPD and why they appear to pose a higher risk subgroup of AMD. So, the collective national and international minds in this study cover a broad range of expertise including bioinformatics, artificial intelligence (AI), genetics, molecular biology, anatomy, stem cell research and clinical ophthalmology.
The study was awarded funding through the Australian National Medical Health and Research Council’s Synergy grants programme in 2019 which was specifically designed to fund projects that required a dedicated effort from multiple groups with different expertise. “This is a large, combined effort in AMD research. Understanding what is different about the high-risk group and why this group is more likely to lose vision is the key to preventing it,” said Prof Guymer.
Drusen, RPD and AMD
The early stages of AMD are diagnosed by the presence of drusen. The size and extent of the drusen present in the macula determine the risk of disease progression. Patients with larger drusen (>125μm) are considered to have intermediate AMD and be at a greater risk of progression compared to those with smaller drusen. With the advent of spectral-domain optical coherence topography (OCT) imaging techniques it is now possible to distinguish more accurately different deposits at the macula. Imaging with OCT distinctly locates RPD above the retinal pigment epithelium (RPE). RPD have a distinct appearance on SD-OCT and their location above the RPE clearly distinguishes them from conventional drusen which are below the RPE (Fig 1). RPD are often difficult to see on colour fundus photographs and clinical examination and as such are often missed. Development of infra-red reflectance scans and fundus autofluorescence (FAF) technology has helped to distinguish these lesions but OCT-B scans are the best way to visualise these deposits. RPD appears to place people at greater risk of AMD progression, but little is known about what causes RPD deposits or how they affect AMD progression, though they are considered important in disease pathogenesis as they are frequently present in late-stage AMD.
Uncovering the genetic pathways associated with RPD could lead to the identification of the pathological mechanisms that are responsible for RPD development, which will then enable research into therapies targeting key points in the disease pathway, explained Dr Carla Abbott, a senior research fellow at CERA and AMD Synergy grant project manager. The first step however is to learn more about their cause which is the current focus of the research group.
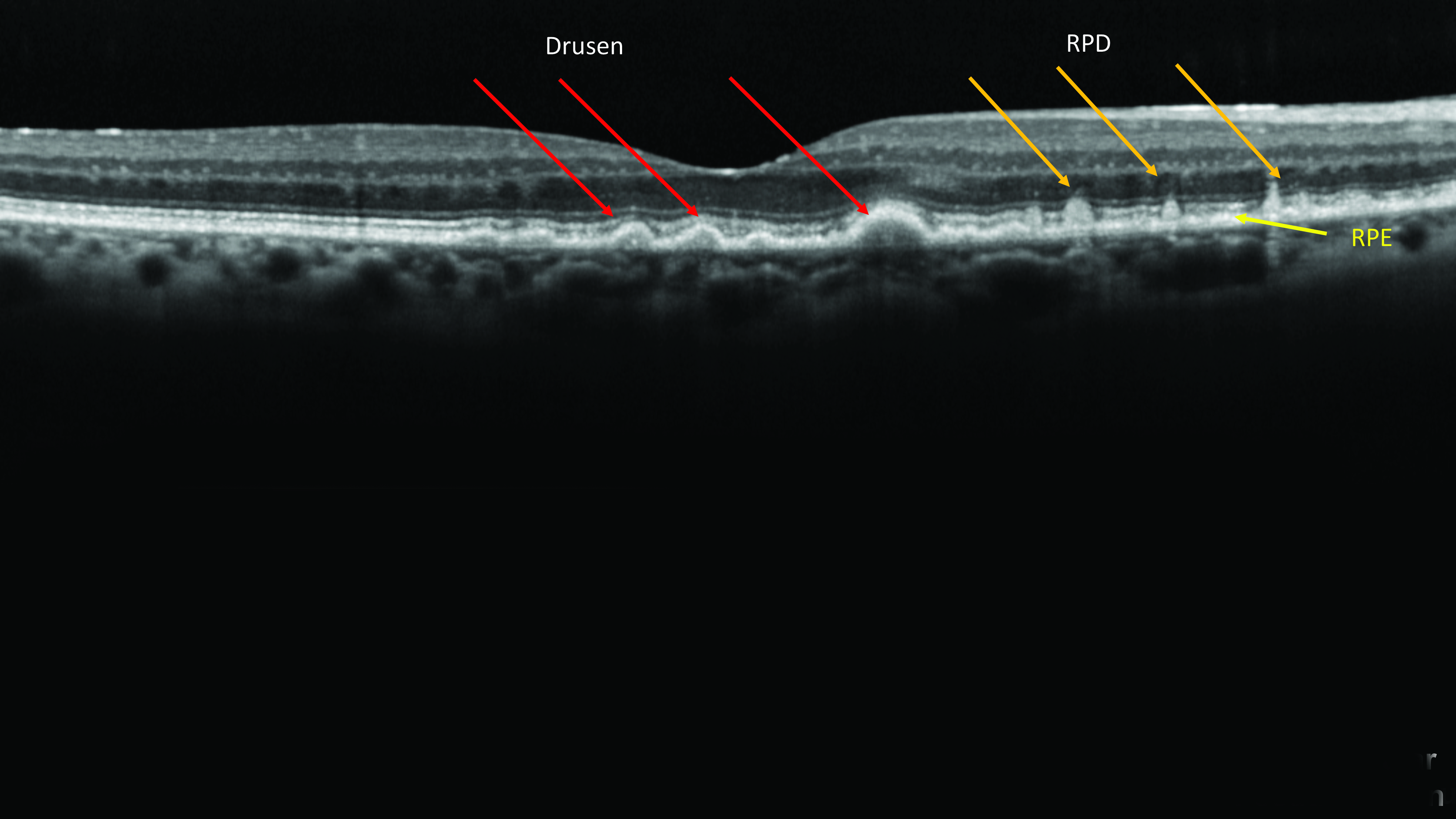
Fig 1. RPD occur on top of the RPE, so are called subretinal drusenoid deposits (SDD)
The LEAD trial
There are, however, multiple AMD research projects underway releasing findings worldwide. CERA’s Laser intervention in early age-related macular degeneration (LEAD) trial revealed how groups of patients responded to subthreshold nanosecond laser treatment. The study ran over three years at five Australian sites and one Northern Ireland site and focused on 300 patients with intermediate AMD. Patients with large drusen and no atrophy were included in the study. Comparison was made between those patients treated with a short pulse nanosecond laser (2RT, AlphaRET, formerly Ellex) and those with a sham treatment. The primary objective was to demonstrate that laser treatment slowed disease progression compared with sham treatment.
Overall findings indicated that the time to late-stage AMD in those treated with laser was not significantly different from the sham group. However when conducting a post-hoc effect modification analysis it was found that in those eyes without the presence of the RPD phenotype there was a four-fold decrease in progression when treated with the laser. Whilst the results in those with RPD suggested a greater disease progression in the treated group compared to the sham group, these results need to be repeated in further large, well-conducted randomised clinical trials. However, if validated, it is the first time that it will have been shown that different AMD phenotypes respond differently to interventions and further raises the importance of RPD in the pathology of AMD. Also, finding out those with RPD did less well with a treatment that targets retinal pigment epithelium (RPE) cells provides some clues as to the status of the RPE in those with RPD, said Prof Guymer. “This suggests that there is greater dysfunction of the RPE in RPD cases, which concurs with other preclinical research being conducted by one of the other investigators in the Synergy grant, Prof Erica Fletcher, from the University of Melbourne. We await further, well conducted randomised studies to determine if 2RT has a place in the treatment of intermediate AMD.”
AMD-RPD online education
To enable eye care professionals to become more familiar with AMD through multi-modal imaging and to improve their ability to more confidently detect RPD, Prof Guymer and her colleague Dr Lauren Ayton from Melbourne University led a project to develop an online learning tool. This tool helps bring eye care professionals up to date with the latest developments in AMD, with a particular focus on how these developments can be brought into daily practise. This self-directed course, AMD for Primary Eyecare Practitioners, focuses on AMD classification, OCT interpretation, including autofluorescence, biomarker identification and more. It also looks at the risk factors for AMD, disease progression, education on intervention for intermediate AMD and new novel therapies for late-stage disease.
As eye care practitioners, we all contribute to the complex AMD puzzle. When it comes to helping our patients preserve their sight, patient and practitioner education about AMD diagnosis and progression is important for prompt referral and initiation of therapy. I, for one, enthusiastically await the results from Prof Guymer’s study. Though this is a five-year project, so a full series of results will not be available for some time, Abbott said the team are hopeful they will be able to share some results through the course of the study.
Louise Wood is a therapeutically qualified optometrist working at City Eye Specialists in Auckland, New Zealand, and a regular contributor to NZ Optics.