Relative afferent pupillary defect: role and assessment
Relative afferent pupillary defect (RAPD) is one of the more important clinical signs in neuro-ophthalmic examination. Quite often its presence is the only objective sign of unilateral or asymmetric dysfunction of the optic nerve. Hence, its detection plays a critical role in making a diagnosis of retrobulbar optic nerve pathology where examination at the slit lamp may not show any abnormality of the ocular media, retina or optic nerve head.
The history of RAPD testing is long, spanning nearly two millennia. Its modern definition and standardisation of assessment, however, is only a few decades old. As far back as 200AD, Galen was using a cover test, ie. exposure of the eye to darkness, to help him to decide whether couching the cataract in that eye would be successful (in other words the eye was healthy besides the cataract) or whether it was irreversibly blind and was best to be left alone1. Galen noticed that in a patient with good vision in both eyes, upon covering one of the eyes with a hand, a small but definite dilation of the pupil occurs in the other eye. The same was true if the eye with cataract, that had good chance of visual improvement after couching, was covered. However, if the blind eye (with or without cataract) was covered, there was no dilation of the other pupil. What Galen observed would, in the 20th century, be termed a “reflex to darkness” but essentially, it was one of the first methods of detecting RAPD. Over the first half of the 20th century, the importance of observing and measuring the difference between pupil sizes, under the conditions of sequential eye cover or pupillary constriction after exposure to bright light, has been further emphasised and different methods described until the “swinging flashlight test” was introduced by Paul Levatin in 1959, when a current way of testing for RAPD was finalised.
Relative afferent pupillary defect arises due to the different amount of sensory input reaching the brainstem from each eye when pupillary constriction is assessed with the alternating light test. The pupillary light reflex is composed of four neurones (Fig 1). The afferent fibres run within the optic nerve and partially decussate at the optic chiasm before entering the midbrain and synapsing with second order neurones in the pretectal nucleus. The second order neurones then synapse with third order neurones in Edinger-Westphal nuclei on both sides. Thus, the sensory input from one eye results in motor output of the same amplitude to both pupils. The efferent fibres leave in the oculomotor nerve and enter the orbit via its inferior division to synapse with final neurone in the ciliary ganglion. Then the fourth order neurone travels via short ciliary nerves to enter the eye and innervate sphincter pupillae muscle.
The alternating light test is employed to detect the presence of RAPD. The test should be done in a dim but not completely dark room. Commonly, a bright source of light from a muscle torch or a direct ophthalmoscope is employed. The dim room and bright, focused light are required to maximise the amplitude of the pupillary movement from a dilated state to fully constricted one, thus making a change in pupil size more obvious for the examiner. A patient is instructed to look at a target in the distance, which will prevent pupillary constriction due to accommodation and therefore ensure larger amplitude of pupillary movement during its exposure to light.
When the eye is illuminated, the light source is held about 20cm away from patient’s face and shown directly onto the pupil. It is held there for two to three seconds and then rapidly swung across and shown directly onto the other pupil for the same duration of time. The pupillary reaction of the eye under the light is then observed. The swinging manoeuvre can be repeated two to three times until the examiner is confident in the findings. If there is still doubt, the light source can be removed from the patient’s eyes for a short period of time to let the retinae recover from bleaching. It is important to illuminate each eye for the same amount of time, otherwise uneven duration of illumination can lead to an artefactual RAPD where in reality there is none present.
The RAPD is not an all or nothing phenomenon and can be of different magnitude depending on the severity of the pathology and thus the amount of afferent information reaching the midbrain. It can generally be divided into mild, moderate or severe. In a mild RAPD, when the light is swung from the normal eye to the one with pathology, the illuminated pupil will constrict but to a lesser degree than in the normal eye. There may also be a faster “release” – that is the pupil in the abnormal eye will dilate faster compared to contralateral eye after the initial maximal constriction. In a moderate RAPD, illumination of the pupil in the abnormal eye (following illumination of the normal eye), will not lead to a constriction, instead the pupil will either stay the same size or show mild dilation. In a severe RAPD, the light switching from the normal eye to an abnormal one will lead to a profound dilation of the illuminated pupil.
The main disease entities responsible for RAPD presence are a variety of either unilateral or asymmetric optic neuropathies of different aetiologies, such as infective or inflammatory optic neuritis, non-arteritic ischaemic optic neuropathy, compression of the optic nerve by a tumour, traumatic optic neuropathy or asymmetric glaucoma. Asymmetric or unilateral retinal pathology can also result in RAPD, but it has to be severe and involve either most of the retina or the entire macula.
Examples of retinal pathologies causing RAPD are central retinal artery occlusion, ischaemic central retinal vein occlusion with severe macula oedema, macula-off retinal detachment and very asymmetric severe age-related macular degeneration. Non-retinal conditions capable of producing RAPD include dense amblyopia (only a mild RAPD is usually present) and intracranial lesions involving optic tract. There is also a very small proportion of subjects with normal visual pathway that were demonstrated to have a very mild RAPD, mostly detected using infrared pupillometer in a research setting and this was attributed to the presence of some variability of the afferent visual system.
In the spirit of the old wisdom that ‘a picture is worth a thousand words’, I have created a video together with Professor Helen Danesh-Meyer where assessment technique and RAPD grading (described above) are demonstrated using real clinical cases which could enhance the understanding of the test. The video can be found on YouTube under the title, Assessment of Relative Afferent Pupillary Defect (RAPD), or at the following link: https://www.youtube.com/watch?v=g0bmA2rS-0o
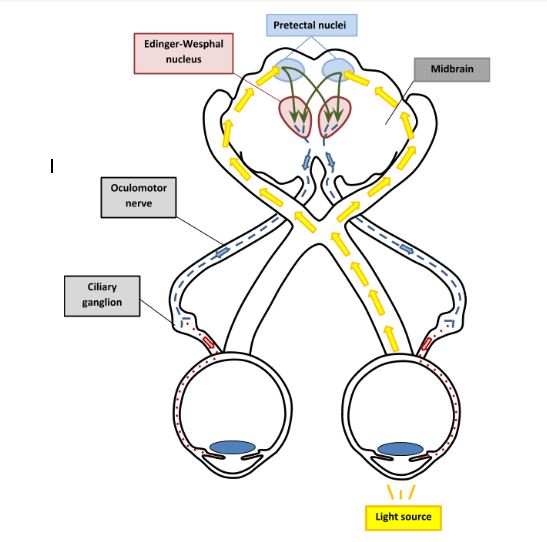
Fig 1. Light reflex pathway. Yellow arrows, first order neurones, green arrows, second order neurones, blue arrows, third order neurones and red arrows, fourth order neurones
About the author
Dr Taras Papchenko is a consultant ophthalmologist at Greenlane Clinical Centre. After finishing his ophthalmology training in New Zealand, he completed specialist training in the UK in oculoplastic surgery and neuro-ophthalmology. He also gained a PhD in ophthalmology (2013) from Auckland University for investigating the effects of laser-induced rodent optic neuropathy on the glia of the optic nerve.
References
Thompson HS. The Vitality of the Pupil: A History of the Clinical Use of the Pupil as an Indicator of the Visual Potential. J Neuro-Ophthalmol, 2003; 23 (3): 213-224.
Lowenstein O, Givner I. Pupillary Reflex to Darkness. Arch Ophthalmol, 1943; 30 (5): 603-609.
Kawasaki A, Moore P, Kardon RH. Variability of the relative afferent pupillary defect. Am J Ophthalmol. 1995; 120 (5): 622-33.
Wilhelm H, Peters T, Ludtke H, Wilhelm B. The prevalence of relative afferent pupillary defects in normal subjects. J Neuroophthalmol. 2007; 27 (4): 263-7.























