Avian eyes vs human eyes: parallels and differences
Birds are maniraptoran theropod dinosaurs that evolved in the Late Cretaceous - 100.5 to 66.0 million years ago (MYA) - their lineage splitting from that of humans around 310–330 MYA. Until 66 MYA dinosaurs dominated the daylight hours, where vision is important, while mammals were furtive, nocturnal and reliant as much on smell as on sight. As a result, most mammals have relatively poor vision but a better sense of smell, while the opposite is true of birds1-3. Humans are somewhat of an exception, however.
Orbit
The orbit of birds differs from that in humans by having only a thin septum interorbitale separating each globe (Fig 1) rather than nose and sinuses and, inferiorly, the arcus jugale, a composite bone attached to the cranial vault via os quadratum4. In mammals these bones have evolved into the small bones of the middle ear.
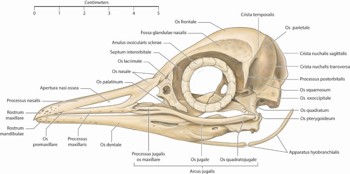
Fig 1. The skull of a little penguin, Eudyptula minor, left lateral view. Note the bony annulus ossicularis sclerae, located in the anterior sclera
Lids
Unlike humans, there is generally more movement in the avian lower lid than in the upper lid and the lid movements are relatively slow as they are moved by smooth rather than skeletal muscle fibres (Fig 2). Some birds only fully close their eyes when they sleep. Instead, the very thin nictitating membrane slides laterally over the globe to clean the cornea, by an action akin to a feather duster.
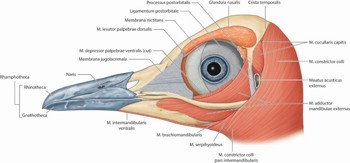
Fig 2. A superficial dissection of the head of a little penguin, Eudyptula minor, left lateral view, showing the structures that surround the eye
As well as a lacrimal gland, producing salty tears for lubrication and a necessary adaptation to terrestrial life, birds also have a Harderian gland that produces an oily sebaceous secretion along the edge of the nictitating membrane.
Globe
Unlike humans, the avian eye is variably aspheric (Fig 3). For instance, owls have a tubular eye to maximise focal length while minimising weight, while the high-acuity eyes of eagles tend to be flat and wide towards the back, maximising image size, but with a variably narrow equator.
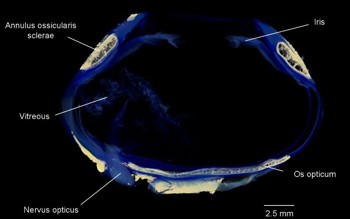
Fig 3. Micro-CT of a king penguin, Aptenodytes patagonicus, eye – note the aspheric
shape of the eye and the bony elements within (beige), the annulus ossicularis sclerae
and the os opticum
Eye movements
The relatively large and aspheric avian globe, fitting snuggly within an orbit that is only slightly bigger than it, limits ocular excursions to such a degree that, in many birds, despite the presence of four rectus and two oblique muscles, the eye is almost unable to move at all. Birds often compensate for this by head movements that are rapid and jerky, ie. saccadic.
Cornea
In humans and birds, the cornea is transparent to ultraviolet C (315–400nm) and visible light. However, there is no sub-basal nerve plexus in birds and the keratocytes are positioned in a more regular array. Rather than anchoring fibrils and plaques, epithelial attachment to Bowman’s layer in birds is by way of incursions. A hexagonal endothelium is present in both, although some are more regular than others. In both, cell density decreases with age5.
The power of the cornea varies between bird species. In general, it increases as the eye becomes smaller, although penguins are notable for having a relatively flat cornea, to reduce the accommodative burden when transitioning from a subaerial to marine environment. For instance, the corneal power in air of the Bald Eagle Haliaeetus leucocephalus is 40D6, very similar to a human’s and much higher than the 19D of the king penguin, despite having a similar axial length of 27mm.
Sclera
In both humans and birds, the sclera is relatively avascular and predominantly collagenous, however, birds have retained a cartilaginous cup around the posterior aspect of the globe (Fig 2), possibly to prevent deformation during accommodation. Birds also possess a ring of overlapping small plates in the anterior sclera. Proposed functions include maintenance of the aspheric shape of the globe, protection, resistance to compression or deformation during extensive accommodation in diving birds and a point of attachment for both intrinsic and extrinsic ocular musculature. Another common bone is the os opticum, around the disc. Not present in all birds, its function is unknown7.
Iris and pupil
In humans and in almost all birds, a healthy pupil is approximately round. However, the king penguin’s becomes square when constricted.
The colour of the avian iris varies widely, from pale blue or yellow to dark brown and may change with age or season. Unlike in humans, both iris sphincter and dilator muscles are composed of striated muscle fibres and the sphincter in particular is well developed. It may be under voluntary control; during avian cataract surgery, intracameral atracurium must be used for dilation, not cyclopentolate.
Lens, accommodation and ciliary body
In humans and birds, ciliary muscle contraction produces accommodation by both forward movement of the lens-iris diaphragm and relaxation of the lens shape. When diving, some birds’ iris sphincter may pinch the anterior lens surface (lenticonus) to further increase the curvature. Some birds can also change the shape of their cornea8.
Humans have excellent spatial resolution and a lens that blocks ultraviolet light (UV). In birds there is a correlation between large eye size, resolution and lack of lenticular UV transmission. Eliminating UV may reduce chromatic aberration, sharpening the image. Furthermore, given the β-peak in the UV range possessed by all visual pigments, all cones are capable of sensing UV light. Thus, UV transmission may make colour differentiation more difficult, an important function in both humans and birds but not in non-primate mammals9.
Vitreous
Unlike in humans, the avian vitreous is firmly attached to the pecten (Fig 4), and a full posterior vitreous detachment is impossible to achieve without cutting it off the pecten.
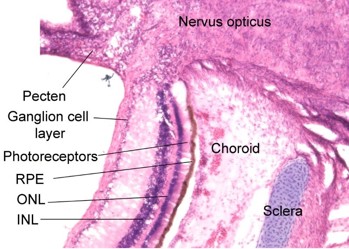
Fig 4. Stained frozen section adjacent to the optic nerve of a seven-week-old gentoo penguin chick, Pygoscelis papua. Note the layered structure of the retina underlain by the retinal pigment epithelium and the cartilaginous component of the sclera, as well as the vascular pecten intruding into the vitreous cavity
Retina
Low-light vision in all vertebrates is mediated by rods, which contain the visual pigment rhodopsin 1.
In humans and birds there is a defined central retinal area dominated by cones and responsible for high spatial resolution. Humans and some birds have a fovea. Other birds have no fovea, two foveae and/or a strip of highly sensitive retina. Some raptors with extremely acute vision have a fovea so steep that it refracts light incident on its slopes, magnifying the image and allowing a visual acuity significantly better than that of humans. Nevertheless, given that humans have a 1:1 correlation between photoreceptors and ganglion cells centrally and a large eye, we do have very good spatial resolution compared with many birds, particularly nocturnal species, which often have greater convergence and greater spatial summation, enabling greater light sensitivity but poorer resolution.
Primates, including humans, are trichromatic. Most birds are tetrachromatic, that is, they have four visual pigments in their colour-differentiating cones. The loss of the rhodopsin 2 in mammals may relate to the period of nocturnal evolution when dinosaurs dominated the daytime. In some birds, short-wavelength-sensitive 1 opsin is maximally sensitive in ultraviolet rather than blue. The peak sensitivity of long-wavelength-sensitive opsin in penguins is shifted to a shorter wavelength, consistent with the preferential attenuation of long wavelength light by water.
Many strongly diurnal birds have ‘double cones’ – a fusion of two cones. They may be useful for achromatic high spatial resolution10. Avian photoreceptor inner segments may contain coloured or clear oil droplets. These may reduce chromatic aberration, protect long wavelength cones against UV damage, act as a waveguide, improve colour perception by spectral filtering and reduce spectral overlap between photopigments, thus improving colour discrimination. However, because they increase light dispersion, they also increase the noise of photoreceptors and impair vision in low light11,12.
Choroid and posterior segment nutrition
Rather than an intrinsic retinal vasculature as in humans, in birds’ eyes inner retinal nutrition is provided by the pecten, a vascular extension into the vitreous of the central retinal artery, and a thicker choroid.
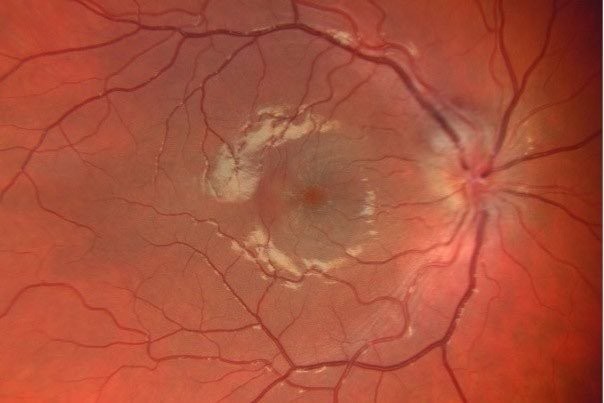
The choroid of diurnal birds tends to be light brown, grey, blue or white (Fig 5). Nocturnal birds (or those that otherwise operate in low light) and humans have a more red or orange fundus13.
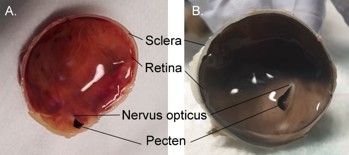
Fig 5. (A) King penguin, Aptenodytes patagonicus (B) gentoo penguin, Pygoscelis papua
Visual fields
The position of the eyes determines the visual field. Forward-facing eyes, as in humans and owls, allows more binocular overlap – likely important in predation and, thanks to our primate ancestors, complex arboreal navigation – while a large brow and lashes avoids direct sunlight on the retina. Conversely, duck eyes are positioned more laterally and not shaded, allowing vision posteriorly and dorsally but limiting binocular overlap, most likely to avoid predation by being able to spot predators in more directions.
Stereopsis
Many birds have their most sensitive part of the retina facing temporally and often choose to examine certain objects with either their left or right eye rather than binocularly. However, binocular foveation is possible in some species, particularly with eye movements.
Whether birds have stereopsis or not is debated. Birds have a completely crossed optic chiasm. A second supraoptic chiasm has evolved to allow about 50% of the nerve fibres to cross the brain a second time. This allows each eye to project to each side of the brain and potentially enables stereopsis. However, birds may use ‘flow-field information’ (ie. the speed at which an object moves across the visual field rather than stereopsis per se) to analyse the time it will take to contact an object14.
References
- Duke-Elder, S. (1958). The eye in evolution: system of ophthalmology, vol. 1. St Louis, London: Mosby, Kimpton. https://doi.org/https://doi.org/10.5962/bhl.title.7241
- Schwab, I. R. (2011). Evolution's witness: how eyes evolved. Oxford University Press. https://doi.org/https://doi.org/10.1111/cxo.12028
- Walls, G. L. (1942). The vertebrate eye and its adaptive radiation. Bloomfield Hills, Mich. : Cranbrook Institute of Science 1942. https://doi.org/https://doi.org/10.5962/bhl.title.7369
- Handbook of avian anatomy: nomina anatomica avium. (1993). (J. J. Baumel, A. S. King, J. E. Breazile, H. E. Evans, & J. C. Vanden Berge, Eds. 2nd ed.). Nuttall Ornithological Club. https://doi.org/https://doi.org/10.2307/1369201
- Hadden, P. W., Gokul, A., Amirapu, S., Kurian, R., McGhee, C. N. J., & Zhang, J. (2023). Confocal and electron microscopic structure of the cornea from three species of penguin. Vision, 7(1), 4. https://doi.org/https://doi.org/10.3390/vision7010004
- Kuhn, S. E., Hendrix, D. V., Jones, M. P., Ward, D. A., Baine, K. H., & Franklin, S. R. (2015). Biometry, keratometry, and calculation of intraocular lens power for the Bald Eagle (H aliaeetus leucocephalus). Veterinary Ophthalmology, 18, 106-112. https://doi.org/https://doi.org/10.1111/vop.12120
- Fischer, O., & Schoenemann, B. (2019). Why are bones in vertebrate eyes? Morphology, development and function of scleral ossicles in vertebrate eyes—A comparative study. J. Anat. Physiol. Stud, 3, 1-26.
- Glasser, A., & Howland, H. C. (1996). A history of studies of visual accommodation in birds. The Quarterly review of biology, 71(4), 475-509. https://doi.org/https://doi.org/10.1086/419554
- Lind, O. M., Mindaugas; Olsson, Peter; Kelber, Almut. (2014). Ultraviolet vision in birds: the importance of transparent eye media. Proceedings of the Royal Society. B, Biological sciences, 281(1774), 1-9. https://doi.org/https://doi.org/10.1098/rspb.2013.2209
- Vorobyev, M., Osorio, D., Bennett, A. T. D., Marshall, N. J., & Cuthill, I. C. (1998). Tetrachromacy, oil droplets and bird plumage colours. Journal of Comparative Physiology, 183(5), 621-633. https://doi.org/10.1007/s003590050286
- Bowmaker, J. (1980). Colour vision in birds and the role of oil droplets. Trends in Neurosciences, 3(8), 196-199. https://doi.org/https://doi.org/10.1016/0166-2236(80)90072-7
- Govardovskii, V. (1983). On the role of oil drops in colour vision. Vision research (Oxford), 23(12), 1739-1740. https://doi.org/https://doi.org/10.1016/0042-6989(83)90192-x
- Wood, C. A. (1917). The fundus oculi of birds: especially as viewed by the ophthalmoscope; a study in the comparative anatomy and physiology. Lakeside Press. https://doi.org/https://doi.org/10.2307/4072242
- Martin, G. R. (2017). The sensory ecology of birds. Oxford University Press. https://doi.org/https://doi.org/10.1093/oso/9780199694532.001.0001

Dr Peter Hadden is an ophthalmologist at Eye Institute and Greenlane Clinical Centre. He is also doing a PhD on penguin eyes at the University of Auckland. His special interests are cataract and vitreoretinal surgery as well as ocular oncology.