Keeping an eye on gut bacteria
In recent years there has been a healthy obsession with what we eat and its influence on our health and wellbeing. According to various studies, gut health has now been linked to the onset of depression, diabetes, poor skin health and much, much more.
The human gut ‘microbiome’ refers to a complex ecosystem of microbial species that colonise the body. It contains up to 100 trillion organisms and plays an important role in maintaining metabolic homeostasis and health through a symbiotic relationship with the host¹. It is largely developed by the time we reach three years of age, although it is modifiable through environmental factors such as diet² throughout life. When the gut microbiota gets out of balance (dysbiosis), however, it can induce systemic inflammation and have a negative effect on our immune system.
The influence of diet
In recent decades the Western diet has evolved with increased consumption of ultra-processed foods and excessive nutrition intake3. Certain foods have been found to cause inflammation in the body, including refined sugars, artificial trans fats, vegetable and seed oils found in processed foods, refined carbohydrates, excessive alcohol and processed meats. Diet profoundly impacts microbial structure in animal models and appropriate gut microbiota in the digestive tract plays an essential role in sending signals to the brain4,5.
The gut microbiota is composed primarily of four bacterial groups: gram-positive Firmicutes (64%), plus gram-negative Bacteroidetes (23%), Proteobacteria (8%) and Actinobacteria (3%)6. The modification of gut composition through the normalisation of its microbiota is a solution for the treatment of gut dysbiosis and could provide novel therapeutic approaches to treat and manage various diseases in different parts of the body, including the eye.
From the immune system to the eye
The immune system benefits from a healthy microbiome as it provides resistance to pathogen colonisation. The gut microbiome has been linked to multiple immune functions, including the production of cytokines, the maintenance of homeostasis and T-cell production. When asked about the relationship between the gut microbiome and its influence on immunity, Professor Clare Wall, head of Auckland University’s Department of Nutrition and Dietetics, says we don’t really know what optimal nutrition is, particularly for developing the microbiome and immunity. “It is a complex area and ongoing research is needed to fully understand the relationship.”
Ageing is associated with a decrease in immune cell signalling and reduced B- and T-cell production. Low-grade inflammation also characterises the ageing process and, as cellular function declines, the risk for age-related diseases increases. For this reason, age-related diseases commonly have an inflammatory pathogenetic origin, termed ‘inflammageing’. Alterations in the gut microbiota have been implicated in a pro-inflammatory state which increases expression of the cytokines interleukin 6 (IL-6) and tumour necrosis factor alpha (TNF-α), which appear to predispose the body to diseases such as Parkinson’s, Alzheimer’s, multiple sclerosis, atherosclerosis, heart disease, diabetes and many more.
Emerging research suggests the existence of a gut-eye axis, wherein gut dysbiosis may be a crucial factor influencing the onset and progression of multiple ocular diseases, including uveitis, dry eye disease (DED), macular degeneration and glaucoma. Furthermore, intestinal microbiota appear to be essential in propagating inflammatory diseases of the eye which could represent a potential target for future therapeutic approaches in the treatment of ocular disease.
Uveitis
Uveitis is generally associated with a systemic infection or immunological disease. More recently the gut microbiota has been implicated in the severity of uveitic disease. Of those who suffer from inflammatory bowel disease, 10% have ocular inflammatory disease (episcleritis, uveitis or conjunctivitis)7. There are many hypotheses to explain the link between uveitis and gut microbiome dysbiosis, including increased gut permeability and decreased immunity leading to the migration of bacterial products or activated immune cells to remote sites and promotion of a pro-inflammatory response8.
Dr James Rosenbaum, chief of the Division of Arthritis and Rheumatic Diseases at Oregon Health & Science University in Portland, says the gut microbiome almost certainly contributes to some forms of uveitis. “In a 2022 study, investigators used cells from the eye and inflamed joints in patients with HLA-B27-associated uveitis and found there was an increase in white blood cells that recognised a protein from bacteria in the gut.”
Patients with uveitis have been found to have decreased levels of several genera of anti-inflammatory bacteria, including Faecalibacterium, Bacteroides and Lachnospira, with increased levels of Prevotella and Streptococcus in their gut microbiome9. Another clinical study found a reduction of gut microbiome diversity in uveitis patients compared with healthy controls10. Many mouse models further support the role of gut microbiota in the onset and severity of autoimmune uveitis11. Although these findings highlight a correlation between gut microbiota and the development of autoimmune uveitis, the underlying mechanism remains unclear.
AMD
The exact pathogenesis of age-related macular degeneration (AMD) remains poorly understood, yet some inflammatory mechanisms associated with innate immunity have been identified. The inflammageing process in AMD gives rise to subretinal drusen which are similar in appearance to irregularities found in drusen in degenerative diseases such as Alzheimer’s and atherosclerosis7. The three layers protecting the retina (inner blood-retina barrier (BRB), outer BRB and the blood-aqueous barrier) undergo changes during ageing, making the retina more susceptible to oxidative stress7.
Obesity is also a proven risk factor for AMD, with one study showing mice fed a high-glycaemic diet (HGD) developed retinal pathology similar to AMD, while the mice on a low-glycaemic diet (LGD) did not12. Furthermore, an HGD has been found to induce gut dysbiosis and an exacerbation of choroidal neovascularisation (CNV) in mice11. These data suggest a high-fat diet plays a role in exacerbating the pathogenesis of CNV by increasing inflammation as a result of gut dysbiosis.
Diabetic retinopathy
Gut dysbiosis has been shown to exacerbate Type 2 diabetes11 and has been associated with prediabetes and type 1 diabetes. A mouse study demonstrated that changing the gut microbiota using intermittent fasting produced beneficial bile acids (namely tauroursodeoxycholate) which provided a neuroprotective effect in the retina and prevented exacerbation of diabetic retinopathy13.
Dry eye
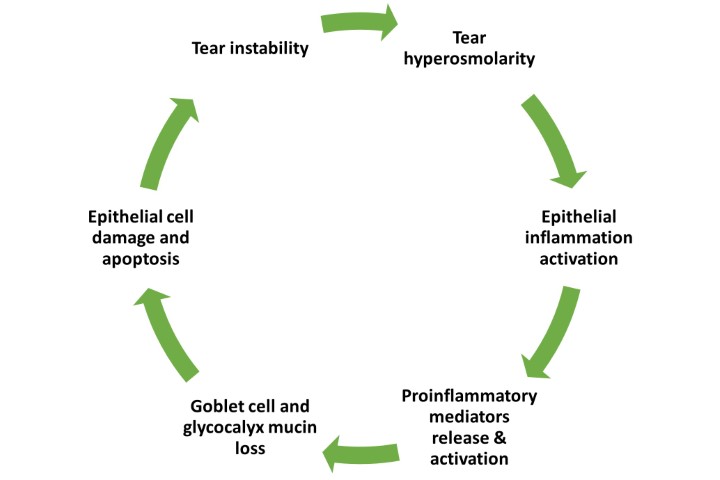
Ocular surface protection is bolstered by a healthy ocular microbiome. Its alteration activates the innate immune system, leading to changes in homeostasis and triggering the DED vicious circle. Emerging research suggests alterations in osmolarity seen in DED and blepharitis could also be related to gut microbiota dysregulation8. “Interestingly, ocular surface mucins and glycoproteins have a similar function to those found in the gastrointestinal tract and the interconnections between mucous membranes throughout the body suggest the gut microbiota may have an impact on various parts of the body, including the eye,” says Associate Professor Maria Markoulli, an optometrist at the School of Optometry and Vision Science at UNSW.
Azadeh Tavakoli, a UNSW PhD student, says there is now strong evidence of gut microbiome dysbiosis in DED patients compared to healthy groups. In animal models, antibiotics used to induce gut dysbiosis have resulted in dry eye10, while restoration of the gut microbiome reversed the DED signs. A study published in 2022 by Tavakoli and colleagues indicated regular consumption of probiotics and prebiotics reduced DED symptoms and improved tear secretion and stability14.
Sjögren syndrome is implicated in dry eye due to lymphocytic infiltration into the lacrimal glands. Modifying the gut microbiome has been shown to modulate the proteins expressed by the lacrimal glands, with IL-10 increasing and IL-1β and IL-6 decreasing10. A study published in 2020 found differences in the gut microbiota in patients with Sjögren syndrome compared with healthy subjects with environmental dry eye syndrome15.
Glaucoma
Research to find a link between gut microbiota and glaucoma is ongoing. A study comparing primary open-angle glaucoma (POAG) patients with healthy individuals found the POAG group had greater levels of Prevotellaceae, Enterobacteriaceae and E. coli and decreased levels of Megamonas and Bacteroides plebeius16. Mean visual acuity was negatively correlated with Megamonas and the mean visual field defect was negatively correlated with the Faecalibacterium genus16. Gut dysbiosis causing chronic and low-grade inflammation may be a trigger for glaucomatous damage, but further research is required to consolidate these findings8.
Improving the gut microbiome
Diet
According to many studies, an increased intake of food filled with ‘good’ bacteria – multicoloured vegetables, leafy greens and coloured fruit – helps boost the microbiome. About 70% of the diet should be raw, organic vegetables, while an increased intake of non-alcoholic fermented foods and drinks, such as kefir, kimchi or kombucha, promotes the population of probiotic bacteria in the gastrointestinal tract. Following a Mediterranean diet – high in fibre-rich foods, lean proteins and healthy fats – also helps promote microbial diversity to positively feed our microbiomes. Intermittent fasting has similarly been reported to have beneficial outcomes, such as decreased insulin, blood pressure, oxidative stress and evening appetite, plus increased insulin sensitivity.
Although he says gut bacteria and diet are incredibly important in triggering uveitis, Dr Rosenbaum says both diet and the make-up of the microbiome are very complex and our understanding of each is far from complete. “I don't believe we currently know enough to recommend a specific diet for everyone with uveitis, but I would avoid extreme changes in diet.” He has at least one patient who believes firmly in eating non-alcoholic fermented foods, such as yoghurt or kimchi, and says there is some emerging science to support that approach. While Dr Rosenbaum has not studied other eye diseases, he says any illness to which the immune system contributes is likely to be affected by bacteria in the intestines.
Foods filled with ‘good’ bacteria – non-alcoholic fermented drinks, multicoloured
vegetables, leafy greens and coloured fruit – help boost the microbiome
Prebiotic/probiotic formulations
Prebiotics (foods, typically high in fibre, which boost the population of specific microorganisms) and probiotics (potentially beneficial microorganisms), help improve your microbiome by promoting the growth of good gut bacteria.
In recent research, Tavakoli, in collaboration with Professor Eric Papas and A/Profs Markoulli and Judith Flanagan, report they found improvements in the signs and symptoms of DED with a combination of prebiotics and probiotics. “The Western diet has been found to be associated with inflammatory conditions in the body. With DED considered an inflammatory disorder, I think modifying the Western diet towards healthier foods can bring about health benefits, maybe even in DED situations,” says Tavakoli.
Emerging research indicates targeting the gut microbiota through prebiotic and probiotic interventions could provide a novel approach to managing and improving the symptoms of DED, agrees A/Prof Markoulli. “The Mediterranean diet has gained attention for its potential benefits in managing chronic inflammatory diseases. In fact, studies have shown individuals who closely follow a Mediterranean diet are less likely to develop primary Sjögren syndrome.”
These findings highlight the potential of using dietary interventions to positively influence the gut microbiome and alleviate DED. Data are still emerging about the use of pre- and probiotics in the treatment of DED and there is currently no guidance regarding the proper dosage, duration or formulation of these supplements.
Faecal microbiome transplantation
Faecal microbiome transplantation (FMT – transferring the gut microbiota of healthy donors into unhealthy recipients) is now widely used to treat Clostridioides difficile infection (CDI), a disease of the large intestine caused by toxins produced by the spore-forming bacterium17. It has been shown to decrease proinflammatory cytokines IL-6 and TNF-α and increase anti-inflammatory bacteria such as Lactobacillaceae and Ruminococcaceae, thus restoring microbiota balance in the recipient17.
“While there is great interest in possible applications of FMT for conditions such as inflammatory bowel disease, autism and obesity, there is no scientific evidence that FMT is safe and effective for treating these problems at this stage,” says Prof Wall. In 2020 a group of researchers at the University of Auckland conducted a trial of gut-microbiome transfer for the treatment of obesity in adolescents. Prof Wall says there was no effect of FMT on weight loss in adolescents with obesity, although a reduction in abdominal adiposity was observed. There’s also no research on FMT and DED, says A/Prof Markoulli.
Conclusion
It will come as no surprise that the gut microbiome is of particular interest in an increasing number of studies investigating eye health. Pathological inflammation induces disruption of the ocular tissues, exacerbating the progression of DR, AMD, uveitis, glaucoma, DED and Sjögren syndrome. It’s also becoming clear that manipulation of the gut microbiome may influence the physiological composition of the ocular microbiome to prevent or even treat eye disease. So by embracing a nutritious eating regime, and encouraging your patients to do the same, you will be supporting your overall health and looking after your eyes.
References
- Siddharth J, Holway N, Parkinson S. A Western diet ecological module identified from the 'humanized' mouse microbiota predicts diet in adults and formula feeding in children. PLoS One. 2013;8(12):e83689.
- Wilson A, Koller K, Ramaboli M, et al. Diet and the human gut microbiome: an international review. Dig Dis Sci. 2020;65(3):723-740.
- Cordain L, Eaton S, Sebastian A, Mann N, Lindeberg S, Watkins B, O'Keefe J, Brand-Miller J. Origins and evolution of the Western diet: health implications for the 21st century. Am J Clin Nutr. 2005 Feb;81(2):341-54.
- González Olmo B, Butler M, Barrientos R. Evolution of the human diet and its impact on gut microbiota, immune responses, and brain health. Nutrients. 2021 Jan 10;13(1):196. PMID: 33435203; PMCID: PMC7826636.
- Martinez K, Leone V, Chang E. Western diets, gut dysbiosis, and metabolic diseases: Are they linked? Gut Microbes. 2017;8(2):130-142.
- Ziętek M, Celewicz Z, Szczuko M. Short-chain fatty acids, maternal microbiota and metabolism in pregnancy. Nutrients. 2021 Apr 9;13(4):1244.
- Scuderi G, Troiani E, Minnella A. Gut microbiome in retina health: the crucial role of the gut-retina axis. Front Microbiol. 2022 Jan 14;12:726792. PMID: 35095780; PMCID: PMC87ak4nfy0k7.
- Napolitano P, Filippelli M, Davinelli S, Bartollino S, dell'Omo R, Costagliola C. Influence of gut microbiota on eye diseases: an overview. Ann Med. 2021;53(1):750-761.
- Kalyana Chakravarthy S, Jayasudha R, Sai Prashanthi G, Ali M, Sharma S, Tyagi M, et al. Dysbiosis in the gut bacterial microbiome of patients with uveitis, an inflammatory disease of the eye. Indian J Microbiol. 2018;58(4):457–69.
- Tavakoli A, Flanagan J. The case for a more holistic approach to dry eye disease: is it time to move beyond antibiotics? Antibiotics (Basel). 2019;8(3):88. Published 2019 Jun 30.
- Floyd J, Grant M. The gut-eye axis: lessons learned from murine models. Ophthalmol Ther. 2020 Sep;9(3):499-513. Epub 2020 Jul 2. PMID: 32617914; PMCID: PMC7406577.
- Rowan S, Jiang S, Korem T, Szymanski J, Chang M-L, Szelog J, et al. Involvement of a gut-retina axis in protection against dietary glycemia-induced age-related macular degeneration. Proc Natl Acad Sci USA. 2017;114(22):E4472–81.
- Beli E, Yan Y, Moldovan L, Vieira C, et al. Restructuring of the gut microbiome by intermittent fasting prevents retinopathy and prolongs survival in db/dbmice. Diabetes. 2018 Sep;67(9):1867-1879. Epub 2018 Apr 30. PMID: 29712667; PMCID: PMC6110320.
- Tavakoli A, Markoulli M, Papas E, Flanagan J. The impact of probiotics and prebiotics on dry eye disease signs and symptoms. J Clin Med. 2022 Aug 20;11(16):4889. PMID: 36013128; PMCID: PMC9409742.
- Moon J, Choi S, Yoon C, Kim M. Gut dysbiosis is prevailing in Sjogren’s syndrome and is related to dry eye severity. PLoS One. 2020;15(2):e0229029.
- Gong H, Zhang S, Li Q, Zuo C, Gao X, Zheng B, et al. Gut microbiota compositional profile and serum metabolic phenotype in patients with primary open-angle glaucoma. Exp Eye Res. 2020;191:107921.
- Lambring C, Siraj S, Patel K, Sankpal U, Mathew S, Basha R. Impact of the microbiome on the immune system. Crit Rev Immunol. 2019;39(5):313-328. PMID: 32422014; PMCID: PMC7362776.

Louise Wood is a therapeutically qualified optometrist working at City Eye Specialists in Auckland and a regular contributor to NZ Optics.